Cell Therapeutics announced on Friday that it will close a XYOTAX drug trial, known as PIONEER, to submit a new trial protocol to the Food and Drug Administration (FDA). The new protocol will be known as PGT306 and will be submitted to the FDA by year’s end.
Because of the particularly low death rate among women patients during the trial, CTIC wants to focus its research on women with normal estogen levels. CEO Dr. James Bianco hopes that this group will be able to increase its uptake and metabolism of the chemotherapeutic drug because of estrogen.
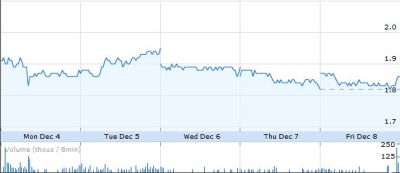
Shares of CTIC dipped 2.2% to $1.86 on 8 December 2006.
Related posts:
Cell Therapeutics To Suspend Enrollment for PIONEER Drug Trial
<URL:http://glob.lokety.com/2006/11/cell-therapeutics-to-suspend.html>
Cell Therapeutics Signs Deal With Norvatis
<URL:http://glob.lokety.com/2006/09/cell-therapeutics-signs-deal-with.html>
Cell Therapeutics Up After Positive Xyotax Phase 1 Results
<URL:http://glob.lokety.com/2006/08/cell-therapeutics-up-after-positive.html>
Cell Therapeutics Signs Financing Agreement
<URL:http://glob.lokety.com/2006/06/cell-therapeutics-signs-financing.html>
Technorati Tags: cell therapeutics, ctic, cancer drug, drug trial, xyotax, pioneer, pgt306, james bianco
